HOW ARE MOST CHEMICAL REACTIONS REVERSIBLE?
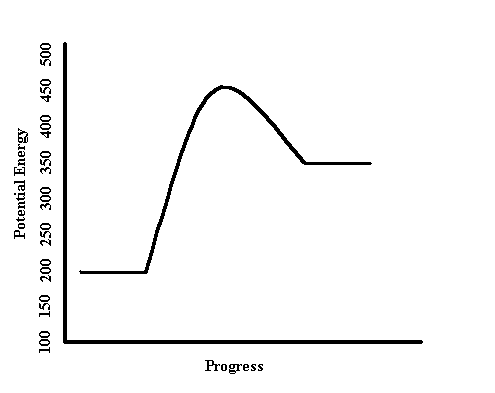
-POTENTIAL ENERGY (PE) DIAGRAM-
The potential Energy diagram above has three main parts. The activation energy (at the top of the 'bump'), the reactants total potential energy (The beginning line), and the products total potential energy (the line at the end of the graph). In order for a reaction to take place, a reactant particle must have an amount of energy equal to or greater than the activation energy. Once this is achieved, the reactant particle will become a product particle, and it's total energy will drop from the activation energy to the product energy. However, this same process can be reversed. If a particle is already in the product state, and it reaches the activation energy, then it will become a reactant particle again, and the energy will drop from the activation energy to the reactant energy. This idea of reversible reactions applies to most chemical reactions.
THE DYNAMIC NATURE OF CHEMICAL EQUILIBRIUM
To understand equilibrium, you have to realize that both forward and reverse reactions are always taking place at the same time. They are just happening at the same pace, and therefore the concentration of a reaction that is at equilibrium will always stay constant. This always changing state of a reaction is why we say 'dynamic' equilibrium - dynamic means 'moving' and chemical reactions at equilibrium are always moving and changing.
WHY WILL A SYSTEM NOT IN EQUILIBRIUM TEND TO MOVE TOWARDS A STATE OF EQUILIBRIUM?
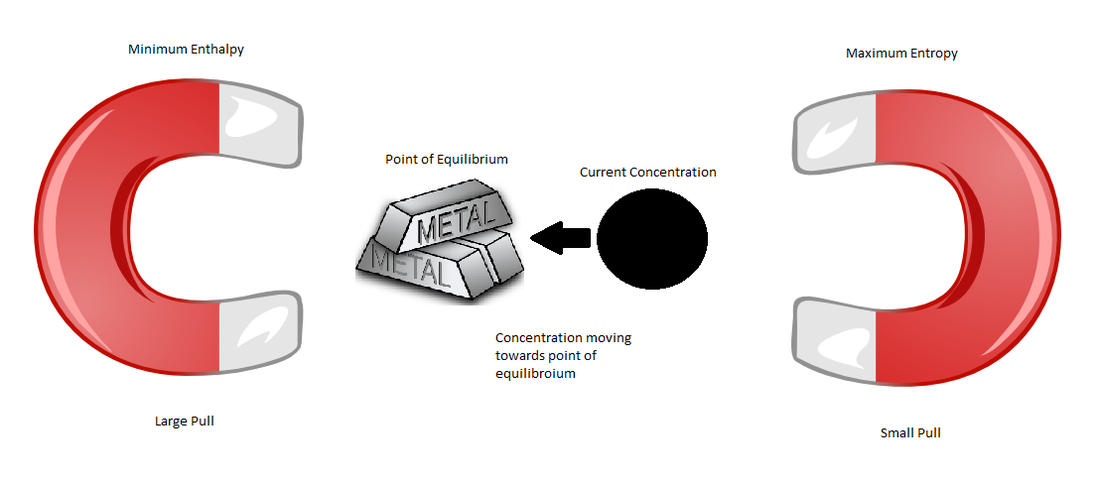
The point of equilibrium in a system is determined by two factors: minimum enthalpy and maximum entropy. Equilibrium is possible when these two factors oppose each other. The exact point of equilibrium is based on the extent of these factors, for instance, if the pull caused by minimum enthalpy is greater than the pull created by maximum entropy, the point of equilibrium will be closer to the side of minimum enthalpy. When the concentration of the particles in the system is not equal to this point of equilibrium, it will shift towards equilibrium because of the attraction of these two factors.
For example, think of these two factors as magnets, with a piece of metal in the middle. Depending on the attraction of these magnets, the piece of metal will be closer to one side or the other.
For example, think of these two factors as magnets, with a piece of metal in the middle. Depending on the attraction of these magnets, the piece of metal will be closer to one side or the other.
CHEMICAL EQUILIBRIUM IN A CLOSED SYSTEM AT A CONSTANT TEMPERATURE WILL HAVE THE FOLLOWING CONDITIONS:
CONSTANT MACROSCOPIC PROPERTIES
Macroscopic properties are properties that are visible to the naked eye (as opposed to microscopic properties, which aren't). The most common macroscopic property that stays constant is the measurement of the concentrations of substances in the reaction. During dynamic chemical equilibrium, as discussed earlier, the microscopic properties are always changing, and reactions are always taking place. However, the rate at which both the forward and reverse reactions are taking place is equal during equilibrium. Therefore to the untrained viewer, it appears that at equilibrium there are no more reactions taking place - this assumption can be described as "during equilibrium the macroscopic properties are constant."
EQUAL FORWARD AND REVERSE REACTION RATES
Imagine two children - child A and child B. Both have 50 transformer toys. Child A has all of his transformer toys in car mode, while child B has all his transformer toys in bot mode. Both children are stealing the others transformer toys, each trying to have the most toys. Each child can only steal one toy at a time from the other child, and must transform the toy before stealing another one. The time it takes to transform a bot into car mode and the time it takes to transform a car into bot mode are equal. Therefore, these children will be stealing each other's toys for all eternity. This is called equilibrium between the children, because both the forward (transforming a bot into a car) and reverse (transforming a car into a bot) processes are taking place at equal rates. The same applies to chemical equilibrium, except instead of transformer toys, it applies to chemicals and elements and chemistry stuff. :D
A REACTION CAN BE ACHIEVED FROM EITHER DIRECTION
Keeping to the idea of transformer toys, keep child A and child B in your head. They are brothers and their mom said child A can have only 25 transformer toys now, while child B can have 75 transformer toys. Currently, all 100 of the transformer toys are in the possession of child A. As hard as child A might try, however, the powers of their mother will prevail until eventually Child A has given up 75 of those toys to child B. If child B had started out with all the toys on the other hand, eventually he would have to give up 25 of them, or else receive a spanking by their mother, who is very strict. To apply this to chemistry, think of the mother as the attractions of minimum enthalpy and maximum entropy, while the transformer toys are different chemicals. No matter how hard the chemicals try, they have no power over the forces caused by the strict enthalpy and entropy. This applies regardless of which chemical in the reaction contains the highest concentration. This is described as "enthalpy can be achieved from either the positive (Child A possessing all the toys) or negative (Child B possessing all the toys) direction.
THE CONCENTRATIONS OF REACTANTS AND PRODUCTS ARE CONSTANT
Since the rate at which the reaction takes place is constant, the concentrations stay constant. If you have ten bananas in a bowl and take away three of them but at the same time your friend puts another three back in, then your concentration is still ten bananas.