ENTHALPY
- the total kinetic and potential energy which exists in a system when it is at a constant pressure -
BUT WHY IS ENTHALPY IMPORTANT?
The Law of Conservation of Energy states that, within a closed system, the total amount of energy remains constant. However, energy may change from kinetic to potential and vice versa. The total energy will not have changed. Enthalpy is a measure of the amount of energy that exists in a system at constant pressure. There are two types of reactions related to enthalpy:
Exothermic - where energy (in the form of heat) is released
Endothermic - where energy (in the form of heat) is absorbed
Reactions tend to move towards the side with the lower enthalpy (minimum enthalpy). On a potential energy diagram, the side with the lower enthalpy is always the side that is lower on the graph. The quantitative value will be lower. In an equilibrium equation, the side with the heat term will be the side with minimum enthalpy.
For example, in the following equation, the products will have minimum enthalpy.
CO(g) + 3H2(g) <=> CH4(g) + H2O(g) + 46.7 kJ
In an endothermic reaction, minimum enthalpy will favour the reactants, while in an exothermic reaction, minimum enthalpy will favour the products.
Exothermic - where energy (in the form of heat) is released
Endothermic - where energy (in the form of heat) is absorbed
Reactions tend to move towards the side with the lower enthalpy (minimum enthalpy). On a potential energy diagram, the side with the lower enthalpy is always the side that is lower on the graph. The quantitative value will be lower. In an equilibrium equation, the side with the heat term will be the side with minimum enthalpy.
For example, in the following equation, the products will have minimum enthalpy.
CO(g) + 3H2(g) <=> CH4(g) + H2O(g) + 46.7 kJ
In an endothermic reaction, minimum enthalpy will favour the reactants, while in an exothermic reaction, minimum enthalpy will favour the products.
ENTROPY
- the amount of randomness in a system -
BUT WHY IS ENTROPY IMPORTANT?
There is a natural tendency for reactions to move to the side with maximum entropy. Reactions that have minimum enthalpy and maximum entropy forces opposing each other will go to equilibrium. Reactions will move to the side with less order.
The side of the reaction equilibrium equation with more moles of gas has maximum entropy.
For example, in the following equation, the reactants will have maximum entropy.
6NH3(g) + 5O2(g) <=> 2NO(g) + 5H2O(g)
Think of a box full of marbles. If it takes you days to organize them completely (order them by colour. size, etc) and your little brother (or sister!) comes along and messes them up a little bit when you aren't in the room, it'll be easy for you to organize them again. It won't take much time to order them again. However, if he messes them up completely, (and there are a lot of marbles!) you will not have the time, patience, or willpower to organize them again. Your lack of will to organize the marbles again are a metaphor for the natural tendencies of reactions. The more random and unorganized the marbles are, the less you will want to organize them again! If fact, it'll be easier to mess them up even more!
Continuing on with the marbles metaphor, think of gases as really little marbles, and solids as really big, gigantic marbles. If your marbles were solids, they'd be much easier to organize than gas marbles!
The side of the reaction equilibrium equation with more moles of gas has maximum entropy.
For example, in the following equation, the reactants will have maximum entropy.
6NH3(g) + 5O2(g) <=> 2NO(g) + 5H2O(g)
Think of a box full of marbles. If it takes you days to organize them completely (order them by colour. size, etc) and your little brother (or sister!) comes along and messes them up a little bit when you aren't in the room, it'll be easy for you to organize them again. It won't take much time to order them again. However, if he messes them up completely, (and there are a lot of marbles!) you will not have the time, patience, or willpower to organize them again. Your lack of will to organize the marbles again are a metaphor for the natural tendencies of reactions. The more random and unorganized the marbles are, the less you will want to organize them again! If fact, it'll be easier to mess them up even more!
Continuing on with the marbles metaphor, think of gases as really little marbles, and solids as really big, gigantic marbles. If your marbles were solids, they'd be much easier to organize than gas marbles!
HOW CAN ENTROPY AND ENTHALPY CHANGES BE DETERMINED FROM A CHEMICAL EQUATION?
There is a natural tendency for a reaction to favour the side with maximum entropy, and minimum enthalpy. The reaction will either go to completion, or not occur at all. However, if they oppose one another, the reaction will go to equilibrium.
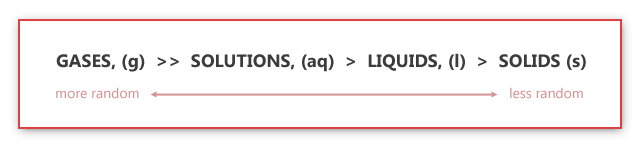
Entropy can be determined from the chemical equation by considering that "Gases > Aqueous > Liquids > Solids". By using this, you can discover whether or not the products or reactants have maximum entropy. However, when both reactants and products have moles of gas, you then count how many moles of gas each side has. (Example above!)
Enthalpy can be determined from the chemical equation by looking to see which side the heat term is on. The side with the heat term is the side with minimum enthalpy. Furthermore, when a forward reaction is endothermic, minimum enthalpy favours the reactants and when a forward reaction is exothermic, minimum enthalpy will favour the products. However, sometimes the heat term is not a part of equation. Oh no! But do not fret! For when there is a will, there is a way! When the heat term is separate from the equation, when it is positive, the forward reaction was endothermic, and the reactants will have minimum enthalpy. Also, when the separate heat term is negative, that means that the forward reaction was exothermic, which means that the products will have minimum enthalpy!
Entropy can be determined from the chemical equation by considering that "Gases > Aqueous > Liquids > Solids". By using this, you can discover whether or not the products or reactants have maximum entropy. However, when both reactants and products have moles of gas, you then count how many moles of gas each side has. (Example above!)
Enthalpy can be determined from the chemical equation by looking to see which side the heat term is on. The side with the heat term is the side with minimum enthalpy. Furthermore, when a forward reaction is endothermic, minimum enthalpy favours the reactants and when a forward reaction is exothermic, minimum enthalpy will favour the products. However, sometimes the heat term is not a part of equation. Oh no! But do not fret! For when there is a will, there is a way! When the heat term is separate from the equation, when it is positive, the forward reaction was endothermic, and the reactants will have minimum enthalpy. Also, when the separate heat term is negative, that means that the forward reaction was exothermic, which means that the products will have minimum enthalpy!
WHEN MINIMUM ENTHALPY AND MAXIMUM ENTROPY BOTH FAVOUR THE PRODUCTS...
- the forward reaction will go to completion , while the reverse reaction will not occur -
Think about this! You have the equation N2O6 <=> 2NO3 + energy. The almighty product side has both more moles of gas than the reactant side, it has the heat term, AND the forward reaction is exothermic! That means that the product side has minimum enthalpy, AND maximum entropy. If above information is correct, which it is, that means that the forward reaction will go to completion, which will make it extremely difficult for the reverse reaction to occur at all. Let's just say that the reverse reaction does not occur!
Think about this! You have the equation N2O6 <=> 2NO3 + energy. The almighty product side has both more moles of gas than the reactant side, it has the heat term, AND the forward reaction is exothermic! That means that the product side has minimum enthalpy, AND maximum entropy. If above information is correct, which it is, that means that the forward reaction will go to completion, which will make it extremely difficult for the reverse reaction to occur at all. Let's just say that the reverse reaction does not occur!
WHEN MINIMUM ENTHALPY AND MAXIMUM ENTROPY BOTH FAVOUR THE REACTANTS...
- the forward reaction will not occur, while the reverse reaction will go to completion -
Riddle me this! You have the equation 2NO3(g) + energy <=> N2O6(g). The reactant side has both more moles of gas than the product side, AND it has the heat term. Which means that the reactant side has maximum entropy and minimum enthalpy. Continuing along with the above logic, the forward reaction will not be able to occur, as the reverse reaction goes to completion.
Riddle me this! You have the equation 2NO3(g) + energy <=> N2O6(g). The reactant side has both more moles of gas than the product side, AND it has the heat term. Which means that the reactant side has maximum entropy and minimum enthalpy. Continuing along with the above logic, the forward reaction will not be able to occur, as the reverse reaction goes to completion.
WHEN MINIMUM ENTHALPY AND MAXIMUM ENTROPY OPPOSE ONE ANOTHER...
- both the forward and reverse reactions will go to equilibrium -
Look! Another example! You have the equation 2NO3(g) <=> N2O6(g) + energy. The reactants have two moles of gas, while the products only have one. Therefore, the reactants have maximum entropy. Furthermore, the forward reaction is exothermic, and the product side has the heat term. Therefore, the product (right) side has minimum enthalpy. As this equilibrium equation has both minimum enthalpy and maximum entropy, the reaction shall go to equilibrium!
Look! Another example! You have the equation 2NO3(g) <=> N2O6(g) + energy. The reactants have two moles of gas, while the products only have one. Therefore, the reactants have maximum entropy. Furthermore, the forward reaction is exothermic, and the product side has the heat term. Therefore, the product (right) side has minimum enthalpy. As this equilibrium equation has both minimum enthalpy and maximum entropy, the reaction shall go to equilibrium!